2008-2009: New Insights into how Neurons Make Dense Core Vesicles
Our work in Adventures>2006 suggested that Dense Core Vesicles (DCVs) were an important component of the synaptic signaling network. However, because DCVs are much less abundant than Synaptic Vesicles (SVs) in the brain, much less was known about their cell biology, and there were some significant mysteries surrounding DCVs. First, DCVs were known to release neuropeptides that modulate behaviors, but it was not known if they had a more essential role for behaviors. For example, blocking synaptic vesicle release completely stops behaviors in C. elegans and other animals. On the other hand knocking out all neuropeptide function can block some behaviors, but other behaviors such as locomotion are much less affected compared to blocking synaptic vesicle function. However, it is important to realize that it was not known if knocking out neuropeptide function was the same as knocking out all DCV functions. In fact, this study showed that knocking out neuropeptide function was not the same as knocking out DCV function. Our study also provided new insights into how DCVs are made. We found that if DCVs lack a protein called Rab2 (or UNC-108 in C. elegans), their maturation is disrupted in a way that doesn't interfere with the neuropeptide releasing function of DCVs, but that does interfere with a second, unidentified function of DCVs.
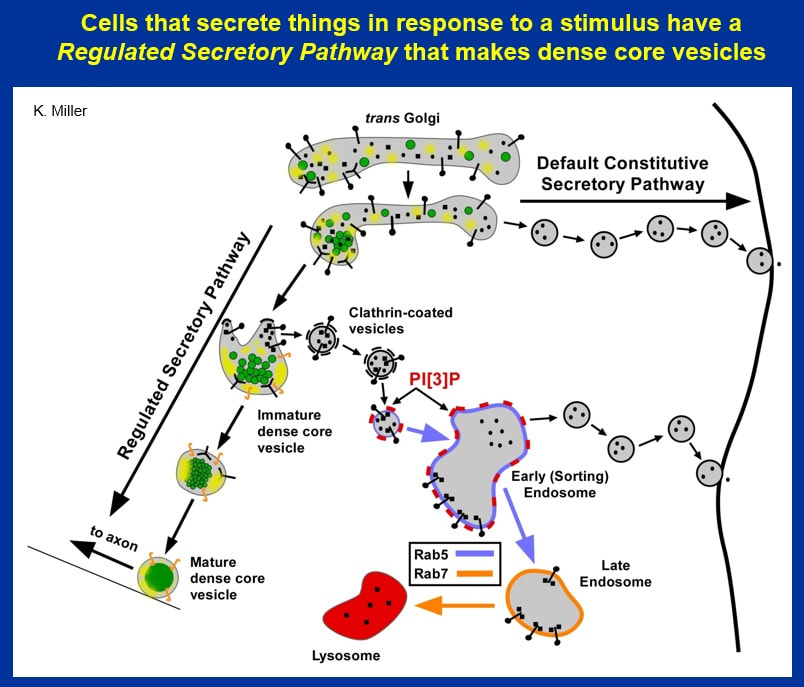
Neurons have a special pathway, known as a "regulated secretory pathway" that makes dense core vesicles. The pathway begins in the trans Golgi (part of the Golgi Apparatus, which is an organelle found in all cells). There, various sorting mechanisms cause Regulated Secretory Proteins, such as neuropeptides, and their processing enzymes to coalescesce and aggregate into vesicles that bud from the trans Golgi to form immature dense core vesicles.
Additional processing and sorting occurs in immature dense core vesicles as the immature DCVs process their neuropeptide cargo(s) and attempt to shed cargos that they don't need.
A PC2 convertase cleaves neuropeptide or secreted protein precursors (pro-neuropeptides or pro-proteins) at dibasic residues. Then carboxypeptidase E trims off the dibasic residues to make the active forms. Neuropeptides have self-aggregating properties, both before and after cleavage. It is not known if larger secreted signaling proteins, such as neurotrophins, which are also packaged into DCVs aggregate like neuropeptides or stay soluble in the vesicle.
In addition to the peptides or secretory proteins that are destined for mature dense core vesicles, immature dense core vesicles have other soluble and transmembrane proteins that were not excluded when the vesicle formed (pinched off) from the trans Golgi.
Studies of Pancreatic beta cells, which secrete the peptide insulin by their regulated secretory pathway, have shown that clathrin-coated vesicles bud from immature dense core vesicles and carry non-dense core vesicle components to early endosomes.
This trafficking event requires the lipid PI[3]P and the Rab protein Rab5. Rab proteins are small GTPases that confer directionality and specificity to vesicle-mediated transport. Rab5 recruits the enzyme that makes PI[3]P and helps target vesicles for fusion with early endosomes.
In this last part of the talk, I’m going to present data showing how this pathway can turn into a bad thing for DCVs and how a different Rab protein, Rab2 (UNC-108 in C. elegans) protects immature DCVs by limiting the activity of this pathway, thus preventing too many soluble and transmembrane proteins from leaving DCVs. If too many soluble and transmembrane proteins leave the immature DCV, a critical non-neuropeptide function of DCVs is disrupted, but the neuropeptide function is not affected, presumably because the neuropeptide remains safely "aggregated" in the center of the forming DCV.
A PC2 convertase cleaves neuropeptide or secreted protein precursors (pro-neuropeptides or pro-proteins) at dibasic residues. Then carboxypeptidase E trims off the dibasic residues to make the active forms. Neuropeptides have self-aggregating properties, both before and after cleavage. It is not known if larger secreted signaling proteins, such as neurotrophins, which are also packaged into DCVs aggregate like neuropeptides or stay soluble in the vesicle.
In addition to the peptides or secretory proteins that are destined for mature dense core vesicles, immature dense core vesicles have other soluble and transmembrane proteins that were not excluded when the vesicle formed (pinched off) from the trans Golgi.
Studies of Pancreatic beta cells, which secrete the peptide insulin by their regulated secretory pathway, have shown that clathrin-coated vesicles bud from immature dense core vesicles and carry non-dense core vesicle components to early endosomes.
This trafficking event requires the lipid PI[3]P and the Rab protein Rab5. Rab proteins are small GTPases that confer directionality and specificity to vesicle-mediated transport. Rab5 recruits the enzyme that makes PI[3]P and helps target vesicles for fusion with early endosomes.
In this last part of the talk, I’m going to present data showing how this pathway can turn into a bad thing for DCVs and how a different Rab protein, Rab2 (UNC-108 in C. elegans) protects immature DCVs by limiting the activity of this pathway, thus preventing too many soluble and transmembrane proteins from leaving DCVs. If too many soluble and transmembrane proteins leave the immature DCV, a critical non-neuropeptide function of DCVs is disrupted, but the neuropeptide function is not affected, presumably because the neuropeptide remains safely "aggregated" in the center of the forming DCV.
|
In Adventures>2005-2007, we performed a huge forward genetic screen in which we screened for suppressors of the hyperactive locomotion and slow growth rate of goa-1 (Go) null mutants. These mutants are slow growing and hyperactive because they have too much Gq pathway activity, and that helped us find the missing Gq effector pathway as described in Adventures>2005-2007. However, we also found many unc-31 mutants in this screen. UNC-31 encodes a protein that is important for DCV exocytosis (fusion and release of its cargos), and it is also critical, directly or indirectly, for proper function of the Gs synaptic signaling pathway, but not for the Gq pathway, as described in Adventures>2006. In addition to unc-31 mutants, we also found other mutants in a gene known as unc-108 that resembled weaker versions of unc-31 mutants. Because of this resemblance to unc-31 mutants, we hypothesized that the unc-108 mutants might disrupt a function related to DCVs.
unc-108 single mutants are relatively sluggish and unresponsive to harsh poking with a wire pick (see videos), but are not paralyzed like an unc-31 null mutant . We showed that we could rescue the sluggish locomotion of an unc-108 mutant by making a transgene that expresses the unc-108 cDNA from its native promoter or from a promoter that drives UNC-108 expression only in neurons. As expected, a transgene with a muscle-specific promoter gave no rescue. unc-108 encodes a protein that is 88% identical to a human protein known as Rab2. There are dozens of kinds of Rab proteins in humans and worms, but Rab2 is the most highly conserved Rab protein in the animal kingdom. To test whether unc-108 and unc-31 mutations disrupt the same pathway/ process, we constructed an unc-108; unc-31 double null mutant. If the unc-108 mutation disrupts the same process as the unc-31 deletion then the locomotion rate of the double should not be any worse than the locomotion rate of unc-31 null, and that is what we found (see paper link below for data). This suggests that the functions of DCVs and Rab2 overlap during locomotion. DCV function in neurons is often equated with neuropeptide release. Pro-neuropeptide processing is required to produce functional neuropeptides. However, as shown on the right, knocking out neuropeptide function with egl-3 and/ or egl-21 mutations results in locomotion rates that are still about 25% of wild type, which is about 8-fold higher than unc-31 null mutants. And yet knocking out both neuropeptide processing on top of an unc-31 null mutation doesn't further worsen the lococomotion phenotype of the unc-31 null. This suggests that neuropeptides and UNC-31 act in the same genetic pathway for locomotion, but that UNC-31 also has another function in driving locomotion that is unrelated to neuropeptides. |
As noted above, we showed that UNC-108 (Rab2) functions in the same process as UNC-31 for locomotion. But is UNC-108 important for the neuropeptide-releasing function of UNC-31 or for the non-neuropeptide related function? To test this, we constructed unc-108; egl-3 double mutants to simultaneously knock out neuropeptide processing and UNC-108 (Rab2). unc-108 single mutants have locomotion rates similar to egl-3 single mutants which have no neuropeptide function. So if UNC-108 is required for neuropeptide function, unc-108; egl-3 doubles should be no worse than either single mutant. Instead, we found that unc-108; egl-3 doubles were much worse than either single mutant and were not significantly different from the unc-31 null. This suggests that unc-108 (Rab2) mutations disrupt the non-neuroeptide related function of UNC-31. So these data suggest that UNC-108 (Rab2) is important for a DCV function, but that it is not required for neuropeptide function.
|
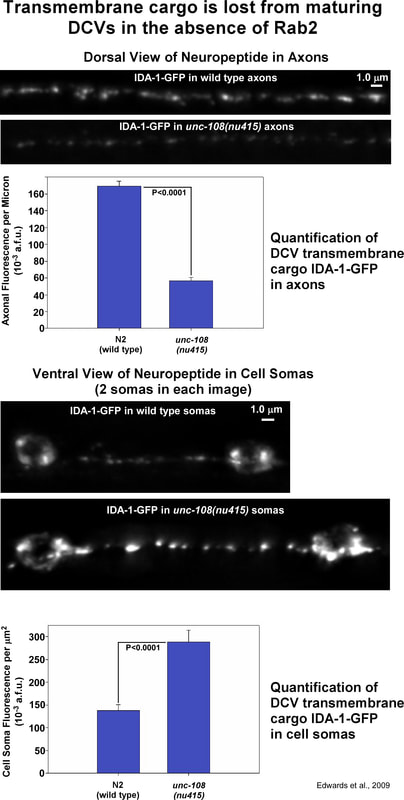
To find out more about how DCV function is disrupted in mutants lacking UNC-108 (Rab2), we made transgenes that tagged various neuropeptides with a fluorescent protein called "Venus" and expressed them in a small subset of motor neurons in the ventral nerve cord. We then crossed those transgenes into unc-108 null mutants, and quantitatively imaged the fluorescently-tagged neuropeptides in the synaptic regions of those neurons on the dorsal side of the animal. The fluorescently tagged neuropeptides show up as white spots (puncta) of various sizes and intensities because they are inside DCVs and the number of DCVs varies between synapses.
For 3 different types of tagged neuropeptides (NLP-21-Venus, FLP-3-Venus, and ANF-Venus), we found that their levels in axons were only about one third of wild type (i.e. two thirds was missing from axons). However, the axonal levels of a 4th neuropeptide, INS-22-Venus was only slightly lower than wild type. One different between the 3 neuropeptides that were lower and INS-22-Venus is that the 3 neuropeptides that were lower all start out as pro-peptides (larger peptides) that are cleaved into smaller functional units by the EGL-3 "pro-peptide convertase". INS-22 contains no EGL-3 cleavage sites, so it is not cleaved. |
An Electron Microscopy study in our paper, performed in collaboration with Janet Richmond's lab, showed that unc-108 null mutants still had the same number of DCVs in axons. So why were the fluorescence intensities of those tagged DCVs in unc-108 mutant axons so much lower than wild type?
Since the fluorescent "Venus" tag was attached to the end of each pro-neuropeptide, cleavage of the neuropeptide by EGL-3 would produce several smaller neuropeptides. Neuropeptides are known to self-aggregate after cleavage while inside DCVs. That is what forms the "Dense Core" of Dense Core Vesicles. However, the Venus that is left after cleavage of the neuropeptides would not be cleaved and would only have a few amino acids attached to it (at its beginning). Thus, the Venus should remain soluble since it has no tendency to self aggregate. We hypothesized that what was lost (or missing) was the soluble Venus cargo, not the aggregated neuropeptide cargo. That would explain why very little of the INS-22-Venus cargo was lost. because INS-22 is not processed into smaller peptide, so the Venus would effectively not be freely soluble and would remain attached to the aggregated INS-22 peptides in the DCV.
Since the fluorescent "Venus" tag was attached to the end of each pro-neuropeptide, cleavage of the neuropeptide by EGL-3 would produce several smaller neuropeptides. Neuropeptides are known to self-aggregate after cleavage while inside DCVs. That is what forms the "Dense Core" of Dense Core Vesicles. However, the Venus that is left after cleavage of the neuropeptides would not be cleaved and would only have a few amino acids attached to it (at its beginning). Thus, the Venus should remain soluble since it has no tendency to self aggregate. We hypothesized that what was lost (or missing) was the soluble Venus cargo, not the aggregated neuropeptide cargo. That would explain why very little of the INS-22-Venus cargo was lost. because INS-22 is not processed into smaller peptide, so the Venus would effectively not be freely soluble and would remain attached to the aggregated INS-22 peptides in the DCV.
If our hypothesis was correct, then there should be normal levels of native neuropeptides in the axons of mutants lacking UNC-108 (Rab2) because most neuropeptide cargos in DCVs are aggregated.
To test this we stained animals with an fluorescent antibody that recognizes a specific class of processed neuropeptides called FMRFamide neuropeptides. We were staining for native neuropeptides in the axons of the dorsal cord. Consistent with our hypothesis, we found that the levels of native FMRFamide neuropeptides in the dorsal cord of unc-108 null mutants was not significantly different from wild type.
To test this we stained animals with an fluorescent antibody that recognizes a specific class of processed neuropeptides called FMRFamide neuropeptides. We were staining for native neuropeptides in the axons of the dorsal cord. Consistent with our hypothesis, we found that the levels of native FMRFamide neuropeptides in the dorsal cord of unc-108 null mutants was not significantly different from wild type.
|
As another test of our hypothesis, we reasoned that we could "rescue" the loss of the soluble Venus cargo from unc-108 mutant axons by blocking peptide processing. To do this, we quantified axonal levels of NLP-21-Venus in the axons of unc-108; egl-3 double mutants (the egl-3 mutation blocks peptide processing). Consistent with the prediction, under these conditions the axonal levels of NLP-21-Venus were actually significantly higher than wild type. The axonal levels of NLP-21-Venus were also higher than wild type in egl-3 single mutants. This indicates that some soluble Venus cargo is lost even in wild type, and that the removal of UNC-108 (Rab2) just worsens a process that occurs even in wild type.
As noted in the drawing of DCV maturation steps near the beginning of this webpage, DCV maturation is thought to occur when clathrin-coated vesicles bud (pinch off) from immature DCVs and carry soluble and transmembrane cargos not destined for mature DCVs to early endosomes. The lipid molecule PI(3)P is essential for the trafficking of membranes through early endosomes. To determine whether the soluble Venus cargo is lost by a PI(3)P-dependent/ early endosome process in unc-108 null mutants, we co-expressed NLP-21-Venus and 2XFYVE-RFP, the latter of which is known to specifically bind to PI(3)P on early endosomes in C. elegans. 2XFYVE has been used in other systems to block the actions of, or neutralize, PI(3)P. Under these conditions, we observed no significant difference between wild type and unc-108 null mutants in the levels or appearance of NLP-21-Venus in axons. These data suggest that the reduced axonal levels of the soluble Venus tag in unc-108 mutants results from its loss by a PI(3)P-dependent process. DCV maturation occurs in neuronal cell somas, not in axons. As shown on the left (2nd image down), the levels of NLP-21-Venus are also reduced in unc-108 single mutant somas. We think this is because the soluble Venus cargo gets taken through the early endosomes and either gets degraded in lysosomes or secreted away. Regardless of whether this happens, blocking neuropeptide processing in unc-108; egl-3 double mutants should restore wild type or greater levels of NLP-21-Venus in cell somas, and that is indeed the case (3rd and 4th images from the top and corresponding graph bars). Similarly, the PI(3)P-dependent loss of the soluble Venus cargo would also be predicted to occur in cell somas during DCV maturation. If this is the case, then there should be no difference in NLP-21-Venus levels in wild type animals with excessive 2XFYVE expression and unc-108 null mutants with excessive 2XFYVE expression, and that is indeed the case (last 2 images and corresponding graph bars). These data thus suggest that the soluble DCV cargo (the Venus in this case) is lost unc-108 mutants, and to a lesser extent in wild type, due to a trafficking event in neuronal cell somas where DCVs under maturation. The clathrin-coated vesicles that carry soluble proteins from immature DCVs to the endosomal system are also thought to carry transmembrane proteins that are not destined for mature DCVs. To investigate the extent to which aberrant trafficking in unc-108 mutants affects the membrane protein composition of mature DCVs, we compared the trafficking of GFP-tagged IDA-1 (IDA-1-GFP) in wild type and unc-108 mutants. IDA-1 is the C. elegans orthologue of a protein that is specific to DCVs in vertebrate neurons and was previously shown to be a marker for DCVs in C. elegans. In wild type, our IDA-1-GFP transgene was associated with a punctate localization in wild type axons similar to NLP-21-Venus expressed in the same neurons. However, crossing the transgene into the unc-108 null mutant reduced the axonal levels of IDA-1-GFP to 33% of wild type, not significantly different from the extent to which NLP-21-derived Venus is reduced in unc-108 mutant axons. However, when we looked at cell somas we observed twice as much IDA-1-GFP as wild type. IDA-GFP is a transmembrane protein with GFP on its cytoplasmic side. We hypothesize that this GFP tag cannot be secreted or easily broken down by lysosomes, so it builds up in the somas after it is removed from the maturing DCVs. In summary, the IDA-1-GFP data suggest that the same process that depletes the soluble Venus tag from the DCV lumen (interior) also strips most of the transmembrane cargo IDA-1-GFP from DCVs as they mature in neuronal somas. |
Edwards SL, Charlie NK, Richmond, JE, Hegermann, J, Eimer, S, and Miller KG. (2009). Impaired dense core vesicle maturation in Caenorhabditis elegans mutants lacking Rab2. J. Cell Biol.186(6):881-895. PMCID: PMC19797080*
*Featured article. See In Focus in same issue: 186(6): 769.
**Co-published with a related article from Stefan Eimer's lab in Germany: Sumakovic, M., J. Hegermann, L. Luo, S.J. Husson, K. Schwarze, C. Olendrowitz, L. Schoofs, J. Richmond, and S. Eimer. 2009. UNC-108/
RAB-2 and its effector RIC-19 are involved in dense core vesicle maturation in Caenorhabditis elegans. J. Cell Biol. 186:897–914.
Remaining Unanswered Questions
One of the most interesting findings from this study was that neuropeptides and UNC-108 (Rab2) separately contribute different parts of the function of UNC-31 (CAPS). The neuropeptide-releasing function of UNC-31 is pretty clear, but we still don't understand the function of UNC-31 that is disrupted in the absence of UNC-108 (Rab2). For the purpose of this discussion, we'll call it the "missing" function of UNC-31, or the "non-neuropeptide-related" function of UNC-31.
UNC-31 had previously been shown to be important for steps preceding the exocytosis of Dense Core Vesicles (DCVs), which is the fusion event that allows them to release their cargo in response to an increase in intracellular calcium. We can't rule out that UNC-31 has a non DCV-related function. Indeed some studies have suggested that it is also involved in synaptic vesicle exocytosis. However, one possibility for the non-neuropeptide-related function of UNC-31 is that it involves the regulated release of other soluble cargos that are not neuropeptides. One such possibility are neurotrophin-like molecules that are known to be released by DCVs. Unlike neuropeptide cargos, which are aggregated, neurotrophin-like cargos are larger signaling proteins that are not known to be aggregated and thus would be susceptible to loss in the absence of UNC-108 (Rab2).
UNC-31 is of special interest to my lab because, in a previous study, we showed that both, or all, of the functions of UNC-31 are required to activate the Gs signaling pathway that is crucial for behaviors, learning, and memory (Adventures>2006), but precisely how UNC-31 connects to the Gs pathway remains unclear.
UNC-31 had previously been shown to be important for steps preceding the exocytosis of Dense Core Vesicles (DCVs), which is the fusion event that allows them to release their cargo in response to an increase in intracellular calcium. We can't rule out that UNC-31 has a non DCV-related function. Indeed some studies have suggested that it is also involved in synaptic vesicle exocytosis. However, one possibility for the non-neuropeptide-related function of UNC-31 is that it involves the regulated release of other soluble cargos that are not neuropeptides. One such possibility are neurotrophin-like molecules that are known to be released by DCVs. Unlike neuropeptide cargos, which are aggregated, neurotrophin-like cargos are larger signaling proteins that are not known to be aggregated and thus would be susceptible to loss in the absence of UNC-108 (Rab2).
UNC-31 is of special interest to my lab because, in a previous study, we showed that both, or all, of the functions of UNC-31 are required to activate the Gs signaling pathway that is crucial for behaviors, learning, and memory (Adventures>2006), but precisely how UNC-31 connects to the Gs pathway remains unclear.
Fun factoids about this study
The C. elegans community tries to be open about what they are working by presenting our work at meetings prior to publication. When Stefan Eimer and I realized that our labs were both working on UNC-108 (Rab2), we colloborate and co-submit our papers. Fortunately, both papers were accepted at the same journal and the articles were published back-to-back.
Stacey Edwards (formerly Stacey Williams; left photo) had only been working for me for 3 years when this paper was published, but this was already her third first author paper. She went on to have many more first author papers and continued working in my lab until it closed in August of 2018. She and Nicky Reynolds (later Nicky Charlie), who worked in the lab from 2000-2006, were extremely hardworking and dedicated and contributed greatly to the success of my lab.
This was the first of several studies in which collaborated with Janet Richmond's lab to add a high pressure freezing electron microscopy analysis of DCVs and/ or SVs in the nerve cord synapses as well as recording synaptic activity through electrophysiology. There are only a few labs and people in the world that can do these two techniques. These methods allow us to see how synapses look and function at the highest possible levels of biological resolution.
Stacey Edwards (formerly Stacey Williams; left photo) had only been working for me for 3 years when this paper was published, but this was already her third first author paper. She went on to have many more first author papers and continued working in my lab until it closed in August of 2018. She and Nicky Reynolds (later Nicky Charlie), who worked in the lab from 2000-2006, were extremely hardworking and dedicated and contributed greatly to the success of my lab.
This was the first of several studies in which collaborated with Janet Richmond's lab to add a high pressure freezing electron microscopy analysis of DCVs and/ or SVs in the nerve cord synapses as well as recording synaptic activity through electrophysiology. There are only a few labs and people in the world that can do these two techniques. These methods allow us to see how synapses look and function at the highest possible levels of biological resolution.
Related Studies
UNC-108 (Rab2) appears to have other functions in non-neuronal cells as highlighted by 2 articles that were published shortly before our article. These other function of Rab2 seem to be related to regulating the mass and/ or lifetime of early and recycling endosomes. Programmed cell death is part of the normal process of development in C. elegans. When a cell dies, it is engulfed (eaten) by a neighboring cell and forms a "phagosome" in the cell that eats it. The phagosome is similar to an early endosome. The phagosome undergoes a "maturation" process in which it slowly transitions to a lysosome, which in turn degrades the dead cell. Two studies found that phagosome maturation is excessively delayed in Rab2 mutants, although it eventually occurs to completion (Lu et al., 2008; Mangahas et al., 2008). One of the studies found that this delay in phagosome maturation correlates with the persistence of the lipid PI(3)P on the surface of the phagosome (Mangahas et al., 2008). Since PI(3)P is a marker for early endosomes, this indicates that the lifetime of early endosomes is increased in Rab2 mutants.
Since our study suggested that DCVs lose their soluble cargo to early endosomes, this may occur simply because early endosomes are sticking around too long, and the normal process of soluble DCV cargo loss to early endosomes during maturation becomes accentuated in mutants lacking Rab2.
I was a co-author on one of these studies because I provided them with unc-108 (Rab2) mutants ahead of publication.
Lu, Q., Zhang, Y., Hu, T., Guo, P., Li, W., and Wang, X. (2008). C. elegans Rab GTPase 2 is required for the degradation of apoptotic cells. Development 135, 1069-1080.
Mangahas, P.M., Yu, X., Miller, K.G., and Zhou, Z. (2008). The small GTPase Rab2 functions in the removal of apoptotic cells in Caenorhabditis elegans. J Cell Biol 180, 357-373.
Another article, this one from Josh Kaplan's lab at Harvard, showed that neurotransmitter receptors, which are normally recycled through early and recycling endosomes in neurons, accumulate at abnormally high levels in the endosomal system. They also reported an increase in tubulovesicular structures that likely correspond to early and recycling endosomes (Chun et al., 2008). This is again consistent with an increase in the lifetime of early/ recycling endosomes that in turn leads to their accumulation in the cell soma affecting processes as diverse as receptor recycling, phagosome maturation, and DCV maturation.
Chun, D.K., McEwen, J.M., Burbea, M., and Kaplan, J.M. (2008). UNC-108/Rab2 Regulates Post-endocytic Trafficking in C. elegans. Mol Biol Cell 19, 2682-2695.
As noted earlier, our paper was co-published with a related study from Stefan Eimer's lab in Germany. They used methods complementary to ours to show that, in UNC-108 (Rab2) null mutants, soluble DCV cargo is lost to endosomes and lysosomes in the cell soma and thus degraded. They also found that RIC-19, which is the C. elegans ortholog of human diabetes autoantigen also acts in the same DCV maturation pathway as UNC-108.
Sumakovic, M., Hegermann, J., Luo, L., Husson, S.J., Schwarze, K., Olendrowitz, C., Schoofs, L., Richmond, J., and Eimer, S. (2009). UNC-108/RAB-2 and its effector RIC-19 are involved in dense core vesicle maturation in Caenorhabditis elegans. J Cell Biol 186, 897-914.
Four studies were published in the years after our paper that identified other proteins that act in the same pathway as UNC-108 (Rab2) for DCV maturation. A study from Tao Xu's lab in China found that a novel, conserved protein called HID-1 acts in the same pathway as Rab2 to prevent mis-sorting of soluble peptide cargos to lysosomes via a PI(3)P early endosome trafficking pathway (Yu et al., 2011). Another study from Stefan Eimer's lab found that TBC-8, a protein necessary to turn Rab2 ON and OFF also functions in the DCV maturation pathway. Then a 2014 paper from Erik Jorgensen's lab at the University of Utah identified two Rab2 interacting proteins that also regulate DCV maturation. Both of these proteins were completely novel, but conserved proteins, thus highlighting the novelty of this new pathway/ process that we helped discover. Finally, a 2016 paper from Michael Ailion's lab at the University of Washington discovered that a protein known as EIPR-1 and other proteins that were part of the EARP complex were also critical components of this pathway. Their paper added further insights by providing evidence that DCV cargo sorting during maturation depends on the EARP-dependent trafficking of soluble cargo through an endosomal sorting compartment.
Yu, Y., Wang, L., Jiu, Y., Zhan, Y., Liu, L., Xia, Z., Song, E., Xu, P., and Xu, T. (2011). HID-1 is a novel player in the regulation of neuropeptide sorting. Biochem J 434, 383-390.
Hannemann, M., Sasidharan, N., Hegermann, J., Kutscher, L.M., Koenig, S., and Eimer, S. (2012). TBC-8, a putative RAB-2 GAP, regulates dense core vesicle maturation in Caenorhabditis elegans. PLoS Genet 8, e1002722.
Ailion, M., Hannemann, M., Dalton, S., Pappas, A., Watanabe, S., Hegermann, J., Liu, Q., Han, H.F., Gu, M., Goulding, M.Q., et al. (2014). Two Rab2 interactors regulate dense-core vesicle maturation. Neuron 82, 167-180.
Topalidou, I., Cattin-Ortola, J., Pappas, A.L., Cooper, K., Merrihew, G.E., MacCoss, M.J., and Ailion, M. (2016). The EARP Complex and Its Interactor EIPR-1 Are Required for Cargo Sorting to Dense-Core Vesicles. PLoS Genet 12, e1006074.
Since our study suggested that DCVs lose their soluble cargo to early endosomes, this may occur simply because early endosomes are sticking around too long, and the normal process of soluble DCV cargo loss to early endosomes during maturation becomes accentuated in mutants lacking Rab2.
I was a co-author on one of these studies because I provided them with unc-108 (Rab2) mutants ahead of publication.
Lu, Q., Zhang, Y., Hu, T., Guo, P., Li, W., and Wang, X. (2008). C. elegans Rab GTPase 2 is required for the degradation of apoptotic cells. Development 135, 1069-1080.
Mangahas, P.M., Yu, X., Miller, K.G., and Zhou, Z. (2008). The small GTPase Rab2 functions in the removal of apoptotic cells in Caenorhabditis elegans. J Cell Biol 180, 357-373.
Another article, this one from Josh Kaplan's lab at Harvard, showed that neurotransmitter receptors, which are normally recycled through early and recycling endosomes in neurons, accumulate at abnormally high levels in the endosomal system. They also reported an increase in tubulovesicular structures that likely correspond to early and recycling endosomes (Chun et al., 2008). This is again consistent with an increase in the lifetime of early/ recycling endosomes that in turn leads to their accumulation in the cell soma affecting processes as diverse as receptor recycling, phagosome maturation, and DCV maturation.
Chun, D.K., McEwen, J.M., Burbea, M., and Kaplan, J.M. (2008). UNC-108/Rab2 Regulates Post-endocytic Trafficking in C. elegans. Mol Biol Cell 19, 2682-2695.
As noted earlier, our paper was co-published with a related study from Stefan Eimer's lab in Germany. They used methods complementary to ours to show that, in UNC-108 (Rab2) null mutants, soluble DCV cargo is lost to endosomes and lysosomes in the cell soma and thus degraded. They also found that RIC-19, which is the C. elegans ortholog of human diabetes autoantigen also acts in the same DCV maturation pathway as UNC-108.
Sumakovic, M., Hegermann, J., Luo, L., Husson, S.J., Schwarze, K., Olendrowitz, C., Schoofs, L., Richmond, J., and Eimer, S. (2009). UNC-108/RAB-2 and its effector RIC-19 are involved in dense core vesicle maturation in Caenorhabditis elegans. J Cell Biol 186, 897-914.
Four studies were published in the years after our paper that identified other proteins that act in the same pathway as UNC-108 (Rab2) for DCV maturation. A study from Tao Xu's lab in China found that a novel, conserved protein called HID-1 acts in the same pathway as Rab2 to prevent mis-sorting of soluble peptide cargos to lysosomes via a PI(3)P early endosome trafficking pathway (Yu et al., 2011). Another study from Stefan Eimer's lab found that TBC-8, a protein necessary to turn Rab2 ON and OFF also functions in the DCV maturation pathway. Then a 2014 paper from Erik Jorgensen's lab at the University of Utah identified two Rab2 interacting proteins that also regulate DCV maturation. Both of these proteins were completely novel, but conserved proteins, thus highlighting the novelty of this new pathway/ process that we helped discover. Finally, a 2016 paper from Michael Ailion's lab at the University of Washington discovered that a protein known as EIPR-1 and other proteins that were part of the EARP complex were also critical components of this pathway. Their paper added further insights by providing evidence that DCV cargo sorting during maturation depends on the EARP-dependent trafficking of soluble cargo through an endosomal sorting compartment.
Yu, Y., Wang, L., Jiu, Y., Zhan, Y., Liu, L., Xia, Z., Song, E., Xu, P., and Xu, T. (2011). HID-1 is a novel player in the regulation of neuropeptide sorting. Biochem J 434, 383-390.
Hannemann, M., Sasidharan, N., Hegermann, J., Kutscher, L.M., Koenig, S., and Eimer, S. (2012). TBC-8, a putative RAB-2 GAP, regulates dense core vesicle maturation in Caenorhabditis elegans. PLoS Genet 8, e1002722.
Ailion, M., Hannemann, M., Dalton, S., Pappas, A., Watanabe, S., Hegermann, J., Liu, Q., Han, H.F., Gu, M., Goulding, M.Q., et al. (2014). Two Rab2 interactors regulate dense-core vesicle maturation. Neuron 82, 167-180.
Topalidou, I., Cattin-Ortola, J., Pappas, A.L., Cooper, K., Merrihew, G.E., MacCoss, M.J., and Ailion, M. (2016). The EARP Complex and Its Interactor EIPR-1 Are Required for Cargo Sorting to Dense-Core Vesicles. PLoS Genet 12, e1006074.